 Therefore, while the Law of Definite Proportions can be used to compare two experiments in which hydrogen and oxygen react to form water, the Law of Definite Proportions can not be used to compare one experiment in which hydrogen and oxygen react to form water, and another experiment in which hydrogen and oxygen react to form hydrogen peroxide (peroxide is another material that can be made from hydrogen and oxygen). The Law of Definite Proportions applies when elements are reacted together to form the same product. The particles were produced by a sample of radium. (a) A representation of the apparatus Rutherford used to detect deflections in a stream of particles aimed at a thin gold foil target. Law of Definite Proportions states that in a given type of chemical substance, the elements are always combined in the same proportions by mass. 7: A Summary of Rutherford’s Experiments. Similarly, when 2 grams of A react with 16 grams of B, they must produce 18 grams of C. 
If 1 gram of A reacts with 8 grams of B, then by the Law of Conservation of Mass, they must produce 9 grams of C. For Thomson’s atomic theory model he was awarded noble prize in 1906 and he died in the year 1940.\): If 1 gram of A reacts with 8 grams of B, then by the Law of Definite Proportions, 2 grams of A must react with 16 grams of B. These negatively charged electron and positively charged proton make an atom neutral. Thomson’s atomic theory model was compared to a Christmas pudding because it was seen that as the currants are embedded in the pudding similarly the negatively charged electrons was also embedded in the positively charged sphere. Refresher: The atomic model originated in the 5th century BC. His work involved the use of cathode ray tubes and identifying a particle lighter than the atom itself, the electron. Thomson discovered that the atom wasn’t as indivisible as previously claimed. What followed was another, shorter lull where our knowledge of atoms didn’t progress all that much. J.J Thomson contributed massively to the model of the atom and the modern day theory. Dalton’s outlining of atomic theory was a start, but it still didn’t really tell us much about the nature of atoms themselves. Thomson named these particles as electrons. The History of the Atomic Model: Thomson and the Plum Pudding. Thomson atomic theory proved that the stream is made up of small particles which is piece of the atom and is negatively charged. Thomson found that the mysterious glowing stream would bend toward a positively charged electric plate. It has been previously seen that if a electric current is passed through a vacuum tube, a steam of glowing material was formed. Thomson discovered electron by the cathode ray tube. Having problem with Alternate Current keep reading my upcoming posts, i will try to help you. The red edible part of a water melon represents the sphere of a positive charge where as the black seeds embedded in it is like the electrons. We can compare the Thomson’s atomic theory model with a water melon. It has no over all negative or positive charge. The positive and the negative charges in an atom are equal in magnitude, due to which an atom is electrically neutral. Thomson, he received a Nobel Prize for his work in physics in 1906. An atom consists of a sphere of positive charge with negatively charged electron embedded in it,Ģ. Joseph John Thomson was an English physicist who made major contributions to atomic theory. Thomson’s atomic theory proposed thatThomson’s atomic theory proposed that:ġ. Is this topic Alpha Decay Equation hard for you? Watch out for my coming posts. Thomson’s atomic theory suggested that the atom is not indivisible as it was of smaller pieces – electrons and protons. In the year 1897 Joseph john Thomson totally changed the view of an atom by discovering electron. 
Till the end of the nineteenth century the concept of atom was similar to a small solid billiard ball. Thomson's atomic theory proposed a model of atom which is known as plum pudding model or Christmas pudding or chocolate chip cookie model. 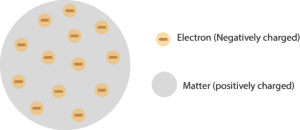 
Thomson atomic theory model came into existence in the year 1903. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
